Background: Chimeric Antigen Receptor (CAR) T-cell therapy is a proven, powerful clinical modality. However, it is still limited by the fundamental obstacle of cancer therapy: discriminating cancer from normal cells. Current FDA-approved CAR T-cell therapies eliminate normal B cells, leaving patients with B cell aplasia, hypogammaglobulinemia, and susceptible to infection.
HLA-Class I loss of heterozygosity (LOH) occurs at an average frequency of ~13% among cancers and specifically ~13% in DLBCL (Broad Institute TCGA database). These losses are irreversible and distinguish the cancer from normal cells. To exploit LOH at the HLA locus, we target the remaining allelic product in tumors with LOH. We evaluated a novel AND NOT Boolean logic gate CAR T module (Tmod) T-cell system to target HLA-A*02 (A2) LOH in lymphoma using both in vitro and in vivo models.
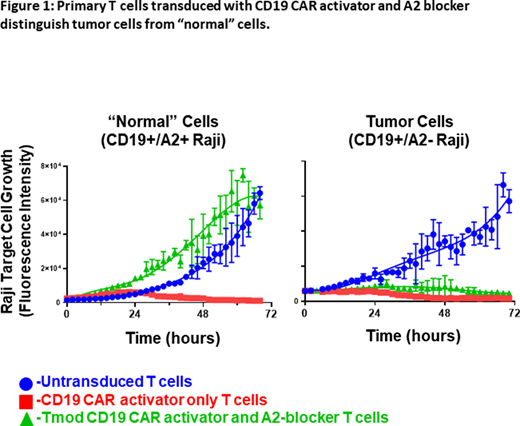
Methods: To model tumor cells that have lost A2 via LOH, we used CD19+ Raji lymphoma tumor cells. To model the corresponding "normal" cells, we established CD19+ Raji cells stably expressing A2 (CD19+/A2+ Raji). We then engineered human primary T cells to express a modular signal-integration circuit designed to be activated only by CD19+ lymphoma that do not express A2 (CD19+/A2- Raji). Each primary Tmod CAR T cell expresses both a CD19 activator (A) module using a CD19-targeting 3rd generation CAR, and a separate A2-targeting blocker (B) module using a novel A2-targeting inhibitory receptor. Human primary Tmod CAR T cells were engineered to co-express the A/B modules. First, T cells were stimulated via CD3/CD28 activation, followed by A/B module lentivirus transduction, and enriched for the B module. In vitro Tmod CAR T cells were evaluated for selective killing of CD19+/A2- Raji compared with CD19+/A2+ Raji. For in vivo proof of concept, both CD19+/A2- Raji and CD19+/A2+ Raji cell lines were injected and established into flanks of immunocompromised NGS mice and challenged with adoptive transfer of engineered human primary Tmod CAR T cells.
Results: Engineered primary Tmod CAR T cells selectively killed CD19+/A2- Raji and spared CD19+/A2+ Raji (Figure 1). Tmod CAR T cells reversibly cycled from a state of non-killing, "block", to cytotoxicity and back, depending on the CD19+/A2- Raji vs. CD19+/A2+ Raji cells to which they were exposed. Importantly, primary Tmod CAR T cells selectively eliminated only the CD19+/A2- Raji cells in mixed cultures. In vivo, Tmod CAR T cells selectively eradicated CD19+/A2- Raji. More importantly, Tmod CAR T cells did not eradicate CD19+/A2+ Raji in vivo.
Conclusions: CD19-targeting Tmod CAR T cells demonstrated robust and selective killing, distinguishing Raji lymphoma lines, one with A2 (CD19+/A2+) and one without (CD19+/A2-), both in vitro and in vivo. A critical requirement for Tmod CAR T-cell therapy is to determine reversibility and lack of anergy in the kill-"block"-kill and "block"-kill-"block" scenarios. This result demonstrates that Tmod CAR T cells do not terminally differentiate into one state (blockade or activation), but rather can switch back and forth as they integrate signals from "normal" and tumor cells. Furthermore, because Tmod CAR T cells can selectively target malignant B cells, it may increase the clinical therapeutic window for CAR T. Tmod CAR T cells may provide a powerful system to address hematologic malignancies and solid tumors with HLA-Class I LOH.
Hamburger:A2 Biotherapeutics: Current Employment, Current equity holder in private company. DiAndreth:A2 Biotherapeutics: Current Employment. Daris:A2 Biotherapeutics: Current Employment, Current equity holder in private company. Munguia:A2 Biotherapeutics: Current Employment, Current equity holder in private company. Deshmukh:A2 Biotherapeutics: Current Employment. Mock:A2 Biotherapeutics: Current Employment, Current equity holder in private company. Asuelime:A2 Biotherapeutics: Current Employment, Current equity holder in private company. Lim:A2 Biotherapeutics: Current Employment, Current equity holder in private company. Kreke:A2 Biotherapeutics: Current Employment, Current equity holder in private company; Gilead: Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months. Tokatlian:A2 Biotherapeutics: Current Employment, Current equity holder in private company. Maloney:A2 Biotherapeutics: Consultancy, Current equity holder in publicly-traded company, Honoraria; Bioline Rx: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Research Funding; Genentech: Consultancy, Honoraria; Gilead Science: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Juno Therapeutics: Consultancy, Honoraria, Patents & Royalties, Research Funding. Go:A2 Biotherapeutics: Current Employment, Current equity holder in private company; Amgen: Current equity holder in publicly-traded company; Allogene: Divested equity in a private or publicly-traded company in the past 24 months; Gilead: Current equity holder in publicly-traded company; Illumina: Divested equity in a private or publicly-traded company in the past 24 months. Kamb:A2 Biotherapeutics: Current Employment, Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal